Led by Yale Cancer Center, research shows that enfortumab vedotin is an effective alternative for patients with muscle-invasive bladder cancer (MIBC)
Presented at the Annual Meeting of the American Society of Clinical Oncology (ASCO) Genitourinary Cancers Symposium (ASCO GU), the Phase 1/b2 EV-103 clinical trial had a number of illuminating findings to share.
“Our findings showed more than one-third of patients displayed no evidence of cancer when their bladder was removed after receiving enfortumab vedotin prior to surgery,” said Daniel P. Petrylak, MD, Professor of Medicine (Medical Oncology) and Urology and Co-Director of the Cancer Signaling Research Program at Yale Cancer Center, and principal investigator for the study.
“The results are significant and so encouraging as survival rates are poor for muscle-invasive bladder cancer.”
Muscle-Invasive Bladder Cancer
In the U.S. bladder cancer is the third most common cancer in men with each year. About 25% of bladder cancers are MIBC, with there being more than 83,000 new cases diagnosed in men and women each year.
MIBC is a type of bladder cancer where the tumour has spread into the muscle of the bladder wall where it is then more likely to spread to other parts of the body. Treatments typically combines cisplatin-based chemotherapy with surgery, however this trial enrolled patients diagnosed with MIBC yet were ineligible for cisplatin-based chemotherapy.
Once enrolled in the trial patients received three cycles of neoadjuvant (prior to surgery) enfortumab vedotin, an anti-body drug, on days one and eight of every three-week cycle.
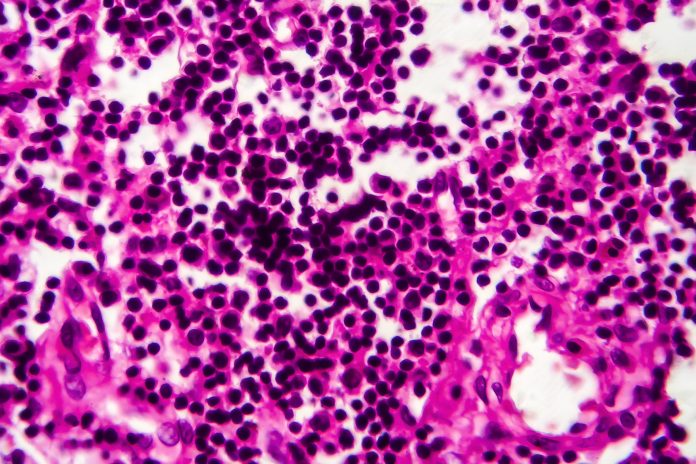
Results from the preliminary analysis of 22 patients showed 36.4 percent had a pathologic complete response, displaying no signs of cancer upon microscopic examination of tissue cells removed during surgery. Half of the patients had a pathological downstaging, or reduction in tumour size.
“We look forward to phase 3 studies to further evaluate the effectiveness of enfortumab vedotin” added Professor Petrylak added.