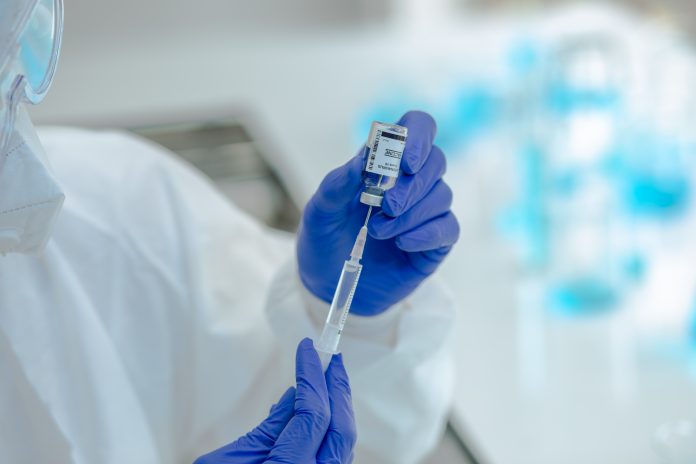
US authorities paused the use of the Johnson & Johnson single dose vaccine, as they investigate six cases of blood clots in women aged between 18 and 48
Health authorities in the United States have temporarily suspended use of the Johnson & Johnson one shot COVID vaccine, as reports of rare blood clots arise.
This situation is similar to the AstraZeneca blood clot investigations in the EU and UK. High-level healthcare bodies like the Center for Disease Control (CDC) and US Food and Drug Administration (FDA) will review data to understand the risk further.
How possible is the blood clot risk?
The FDA describe these events as “extremely rare”, with 6 cases of rare blood clots happening over 7 million Johnson & Johnson jabs in the country.
Currently, the Center for Disease Control (CDC) are reviewing data on six cases of cerebral venous sinus thrombosis (CVST), seen in combination with low levels of blood platelets (thrombocytopenia).
These potential side effects all occurred in women between 18 to 48 years old, with symptoms arising six to 13 days after they got the vaccine.
The FDA ask that: “People who have received the J&J vaccine who develop severe headache, abdominal pain, leg pain, or shortness of breath within three weeks after vaccination should contact their health care provider.”
On Wednesday (14 April), the CDC will have a meeting of the Advisory Committee on Immunization Practices (ACIP) to look over the data.
Similar to possible AstraZeneca risk?
When it came to AstraZeneca, the UK simply adjusted vaccination guidelines – so that under-30s would be required to opt for a different vaccine. The UK now has three approved vaccines, with an expected shortfall in AstraZeneca supply making the decision relatively harmless to their vaccination timeline.
The European Medicines Agency ruled that AstraZeneca remained safe to use, but several countries in the EU created stricter restrictions as a response.
The discussion and public condemnation by various countries shook public confidence in the AstraZeneca vaccine. The use of misinformation about vaccinations can prevent vulnerable individuals from gaining protection against COVID-19.