An experimental Alzheimer’s drug called donanemab, developed by Eli Lilly, has demonstrated a slowdown in progression of disease at early stages, according to trial data
The experimental Alzheimer’s drug reduced cognitive decline by nearly double the reported rate for the overall treatment group.
However, the study revealed less robust results for older patients in later stages of the disease and those with higher levels of the protein tau, which is associated with Alzheimer’s progression.
Earlier detection is crucial in fighting Alzheimer
Anne White, President of Neuroscience at Lilly, emphasised that the findings underscore the importance of early detection and diagnosis in altering the trajectory of Alzheimer’s disease.
The study revealed that patients in the earliest stages experienced a 60% reduction in disease progression, highlighting the potential impact of early intervention using the experimental Alzheimer’s drug.
Side effects of experimental Alzheimer’s drug
Brain swelling, a known side effect of drugs like donanemab, occurred in over 40% of patients with a genetic predisposition to develop Alzheimer’s.
Previously, Lilly reported brain swelling in 24% of the overall donanemab treatment group.
Additionally, brain bleeding occurred in 31% of the donanemab group and approximately 14% of the placebo group. The study reported three trial patients’ deaths linked to the treatment.
However, most of these side effects were manageable through monitoring with magnetic resonance imaging (MRI) or discontinuing the drug.
Donanemab’s mechanism and treatment effect
Donanemab, similar to Leqembi, a drug developed by Eisai and Biogen recently approved for Alzheimer’s treatment, is an intravenous antibody designed to eliminate deposits of beta-amyloid protein from the brains of Alzheimer’s patients.
The trial demonstrated that donanemab’s treatment effect continued to increase relative to the placebo group throughout the 18-month duration, even when participants ceased taking the drug after significant reductions in amyloid deposits.
These findings support the notion that Donanemab can be discontinued once amyloid is cleared from the brain.
Potential for slowing down Alzheimer’s progression
Dr Liz Coulthard, associate professor in dementia neurology at the University of Bristol, expressed optimism about the study’s potential impact on treating Alzheimer’s.
She stated that within the next year or two, patients may have a choice of treatments that slow down the progression of the disease.
Some patients experienced minimal deterioration during the trial; on average, the disease’s advance was delayed by 4.4 to 7.5 months over 18 months.
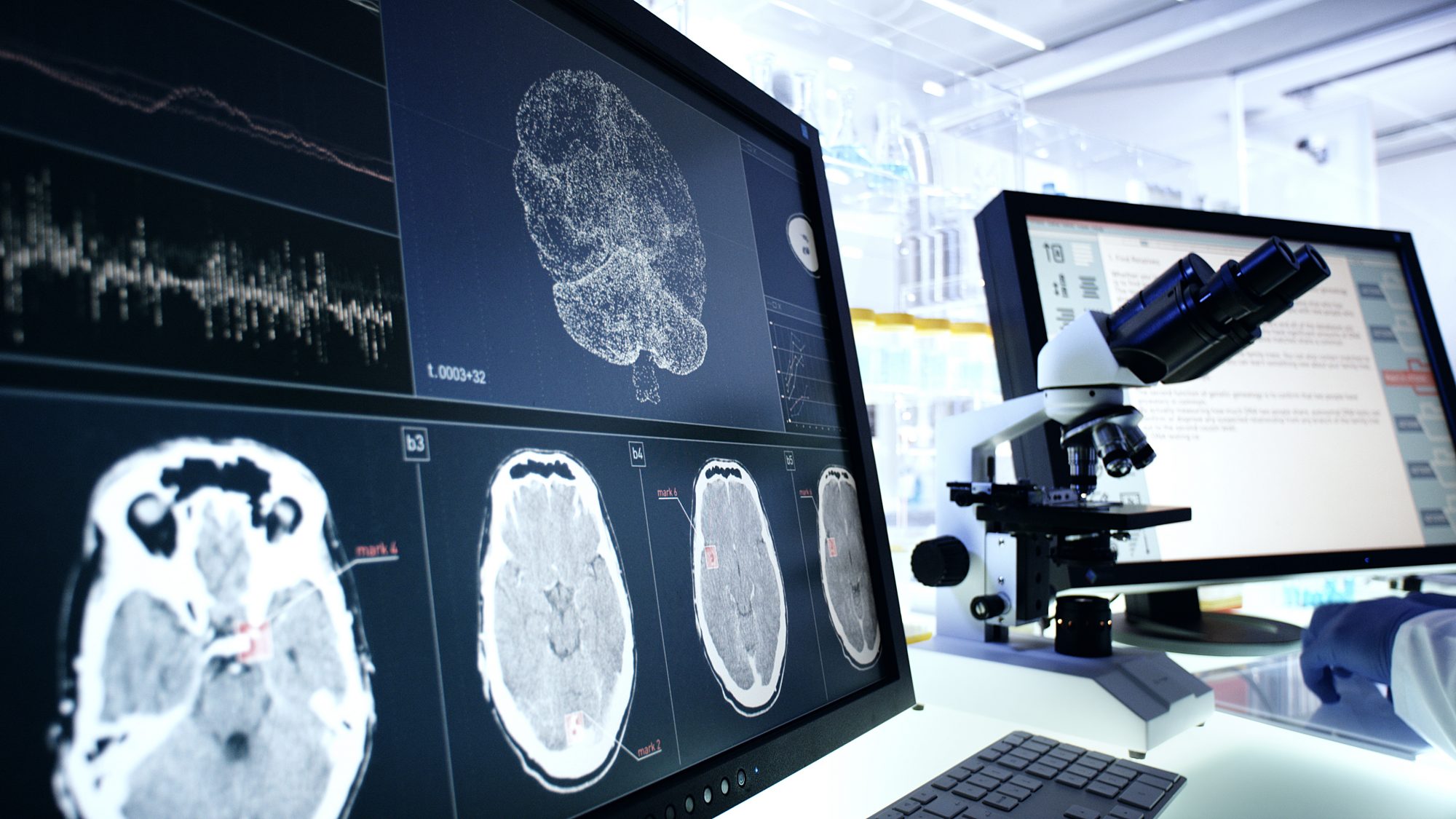
Further evaluation needed for experimental Alzheimer’s drug
An editorial accompanying the study in JAMA highlighted the need for more data to determine whether the modest clinical benefits of these drugs outweigh the potential harms.
The study results were presented at the Alzheimer’s Association International Conference in Amsterdam and published in JAMA.
Regulatory approval and future outlook
Lilly anticipates the U.S. Food and Drug Administration (FDA) ‘s decision to approve donanemab by the end of this year. The company has also initiated submissions to other global regulators, with most expected to be completed by year-end.
This month, the FDA granted standard approval to Leqembi, marking the first disease-modifying treatment for Alzheimer’s to achieve this milestone and paving the way for broader insurance coverage.
Both donanemab and Leqembi are also being studied in large trials to evaluate their potential to delay the onset of Alzheimer’s symptoms.
Alzheimer’s prevalence and stock performance
According to the Alzheimer’s Association, more than 6 million Americans live with Alzheimer’s disease, and projections suggest this number could reach nearly 13 million by 2050.
In early trading on the New York Stock Exchange, shares of Lilly, which reached an all-time high of approximately $469 in June, experienced a slight decline of half a percentage point, trading at $447.28.